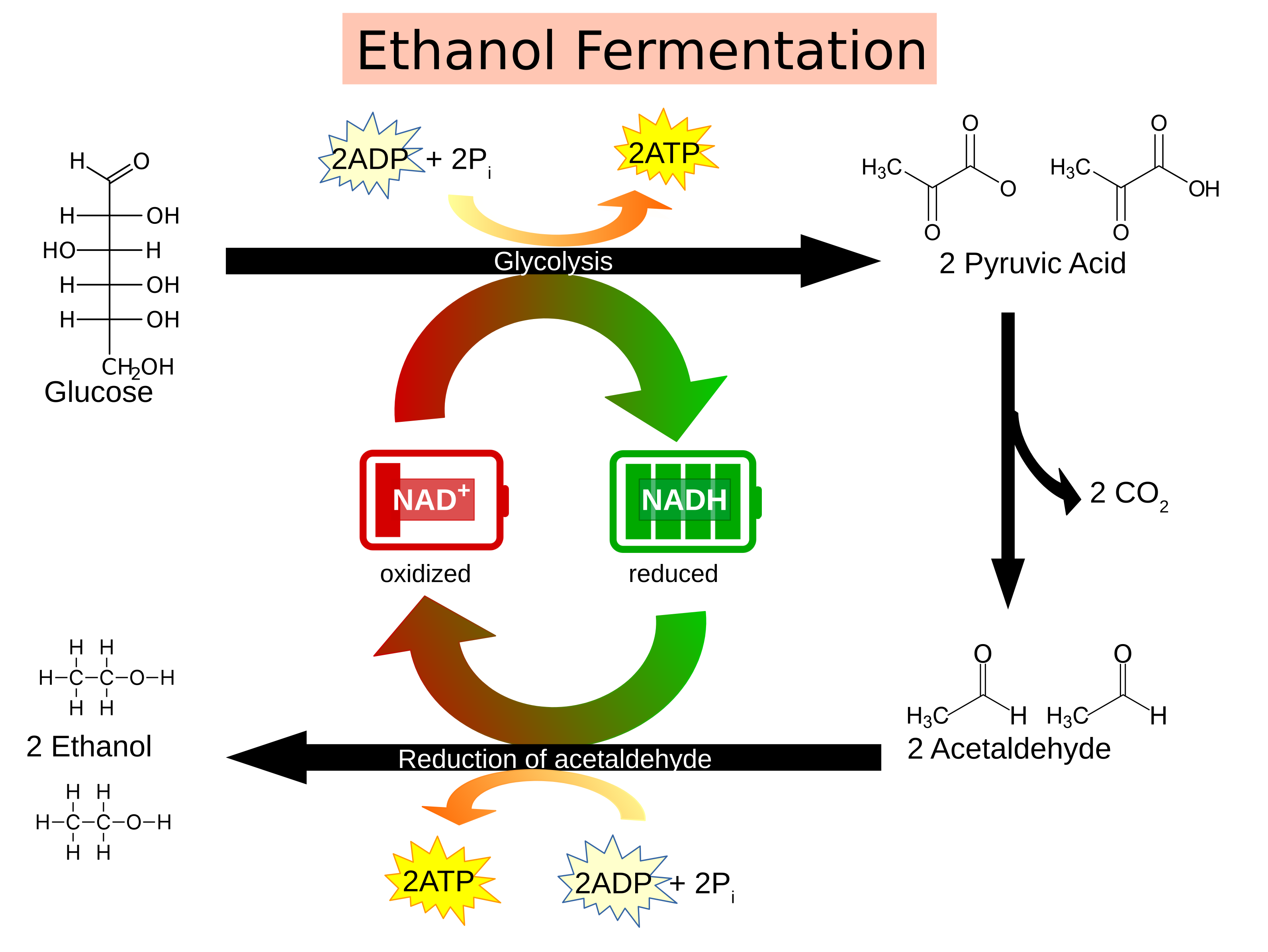
Yeast are single-celled fungi. The species called Saccharomyces cerevisiae is commonly called Baker’s or Brewer’s yeast. Like other eukaryotes with mitochondria, yeast can use oxygen to generate ATP in the process of oxidative phosphorylation. These yeast are facultative aerobes which means they can also switch to an anaerobic mechanism of ATP production called fermentation. In all organisms, the process of glycolysis occurs anaerobically in the cytoplasm to produce two pyruvate molecules from a single glucose. This process produces 2 new ATP molecules and reduced nicotinamide adenine dinucleotide (NADH).
Fermentation is an anaerobic process that occurs in the cytoplasm and quickly generates an additional ATP through the reduction of pyruvate. NADH is the source of electrons in this process that is oxidized to NAD+. Many organisms will ferment to generate lactic acid and CO2 from the pyruvate in order to generate ATP. Yeast fermentation produces ethanol.
Contents
Fermentation Set-up
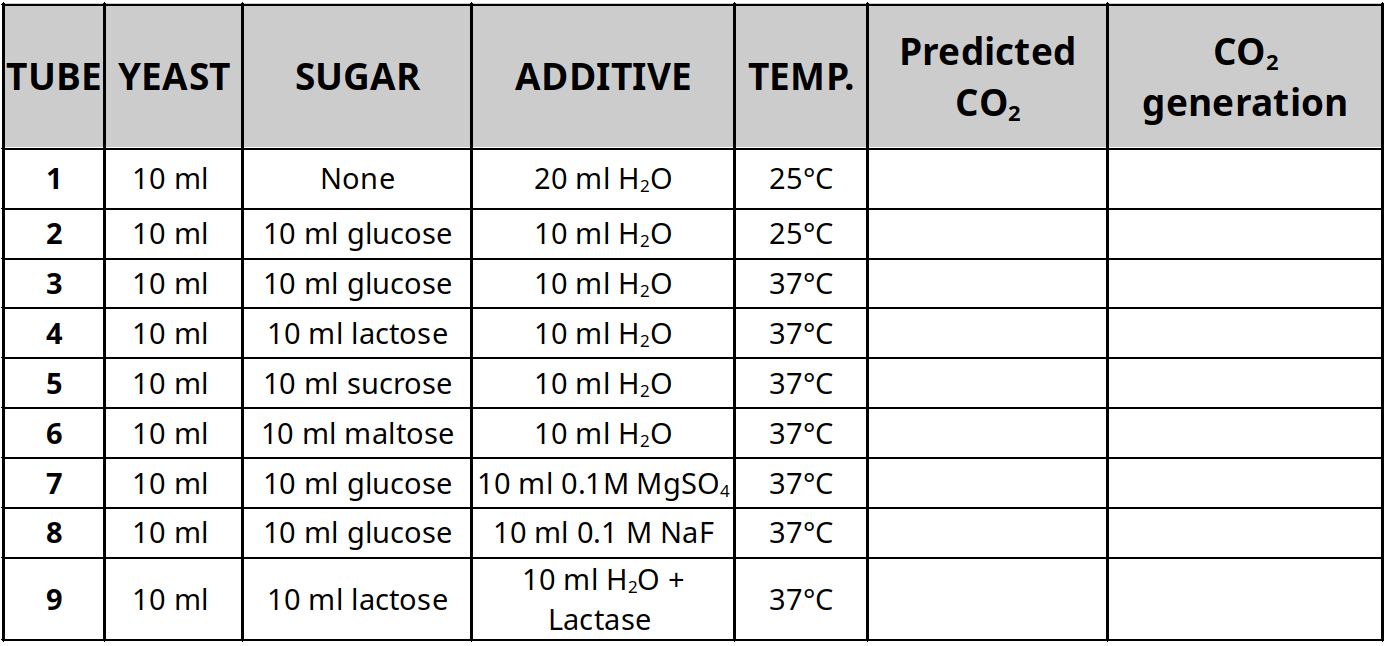
- Mix the solutions in the table below in a fermentation tube
- Eliminate the air bubble in the sealed end of the fermentation tube
- Place rubber stopper into the open end of the fermentation tube and place at the appropriate temperature for an hour
- Predict the amount of CO2 generated in the last column of the table using +, – or +++
after an hour, measure the head space created by the bubbles and compare with your predictions
Questions to direct hypothesis formation
- What is the preferred energy source of the all cells?
- What types of sugars are being used in each tube (monosaccharide, disaccharide, etc)?
- What effect should temperature have on the fermentation reactions?
- What does CO2 indicate in these tubes?
- What do you think the additives do? What effect will they have?